![In dreptunghiul ABCD, cu AB=[tex]8 \sqrt{3} [/tex]și AD=24cm, se duce înălțimea DE a triunghiului ADC, - Brainly.ro In dreptunghiul ABCD, cu AB=[tex]8 \sqrt{3} [/tex]și AD=24cm, se duce înălțimea DE a triunghiului ADC, - Brainly.ro](https://ro-static.z-dn.net/files/d75/c9775ac819b525b2f7887be27ce511f5.jpeg)
In dreptunghiul ABCD, cu AB=[tex]8 \sqrt{3} [/tex]și AD=24cm, se duce înălțimea DE a triunghiului ADC, - Brainly.ro
![1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems - ScienceDirect 1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0022326322014803-jo1c01376_0013.jpg)
1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems - ScienceDirect

gem‐Dihalocyclopropanes as Building Blocks in Natural‐Product Synthesis: Enantioselective Total Syntheses of ent‐Erythramine and 3‐epi‐Erythramine - Stanislawski - 2007 - Chemistry – An Asian Journal - Wiley Online Library

Enantioselective Total Synthesis of (−)-Caldaphnidine O via a Radical Cyclization Cascade | Journal of the American Chemical Society

Evolutions of precision radical polymerizations from metal-catalyzed radical addition: living polymerization, step-growth polymerization, and monomer sequence control | Polymer Journal

Evolutions of precision radical polymerizations from metal-catalyzed radical addition: living polymerization, step-growth polymerization, and monomer sequence control | Polymer Journal

Evolutions of precision radical polymerizations from metal-catalyzed radical addition: living polymerization, step-growth polymerization, and monomer sequence control | Polymer Journal

learning task 1 A. simplify the radical expressions 1.√63 2.√48 3.√75 4.√99 5.√92 6.∛24 7.∛81 8. - Brainly.in
![1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems - ScienceDirect 1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0022326322014803-jo1c01376_0008.jpg)
1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems - ScienceDirect

Diastereoselective Radical 1,4-Ester Migration: Radical Cyclizations of Acyclic Esters with SmI2 | Journal of the American Chemical Society
![1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems | The Journal of Organic Chemistry 1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/acs.joc.1c01376/asset/images/medium/jo1c01376_0009.gif)
1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems | The Journal of Organic Chemistry
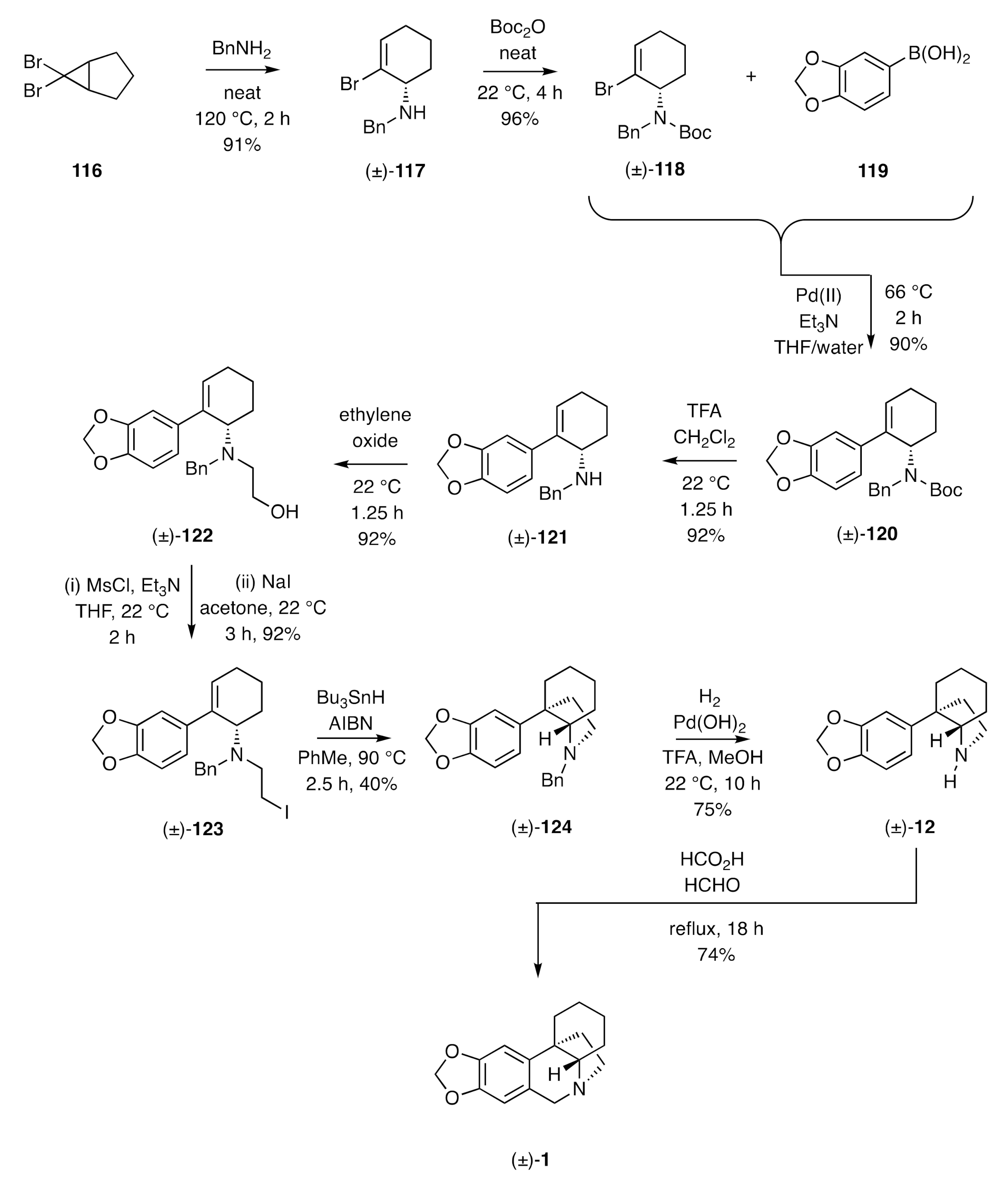
Molecules | Free Full-Text | The Chemical Synthesis of the Crinine and Haemanthamine Alkaloids: Biologically Active and Enantiomerically-Related Systems that Serve as Vehicles for Showcasing New Methodologies for Molecular Assembly | HTML

Synthesis of the Tetracyclic ABCD Ring Domain of Calyciphylline A-Type Alkaloids via Reductive Radical Cyclizations | Organic Letters

gem‐Dihalocyclopropanes as Building Blocks in Natural‐Product Synthesis: Enantioselective Total Syntheses of ent‐Erythramine and 3‐epi‐Erythramine - Stanislawski - 2007 - Chemistry – An Asian Journal - Wiley Online Library
![Catalytic [2+2+2] Tandem Cyclization with Alkyl Substituted Methylene Malonate Enabling Concise Total Synthesis of Four Malagasy Alkaloids | CCS Chem Catalytic [2+2+2] Tandem Cyclization with Alkyl Substituted Methylene Malonate Enabling Concise Total Synthesis of Four Malagasy Alkaloids | CCS Chem](https://www.chinesechemsoc.org/cms/asset/0002b2e9-fa0e-4995-9e88-83ffdf85a72c/keyimage.jpg)
Catalytic [2+2+2] Tandem Cyclization with Alkyl Substituted Methylene Malonate Enabling Concise Total Synthesis of Four Malagasy Alkaloids | CCS Chem

Companie Democraţie Nedreptate in cubul abcda'b'c'd o ac bd.aratati ca a'c abc membrelor printre hardware
![1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems - ScienceDirect 1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0022326322014803-jo1c01376_0012.jpg)
1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems - ScienceDirect

dacă un dreptunghi ABCD are diagonala AC=16 radical din 3 cm și AD=12 radical din 5 cm, atunci aria - Brainly.ro

Diastereoselective Radical 1,4-Ester Migration: Radical Cyclizations of Acyclic Esters with SmI2 | Journal of the American Chemical Society
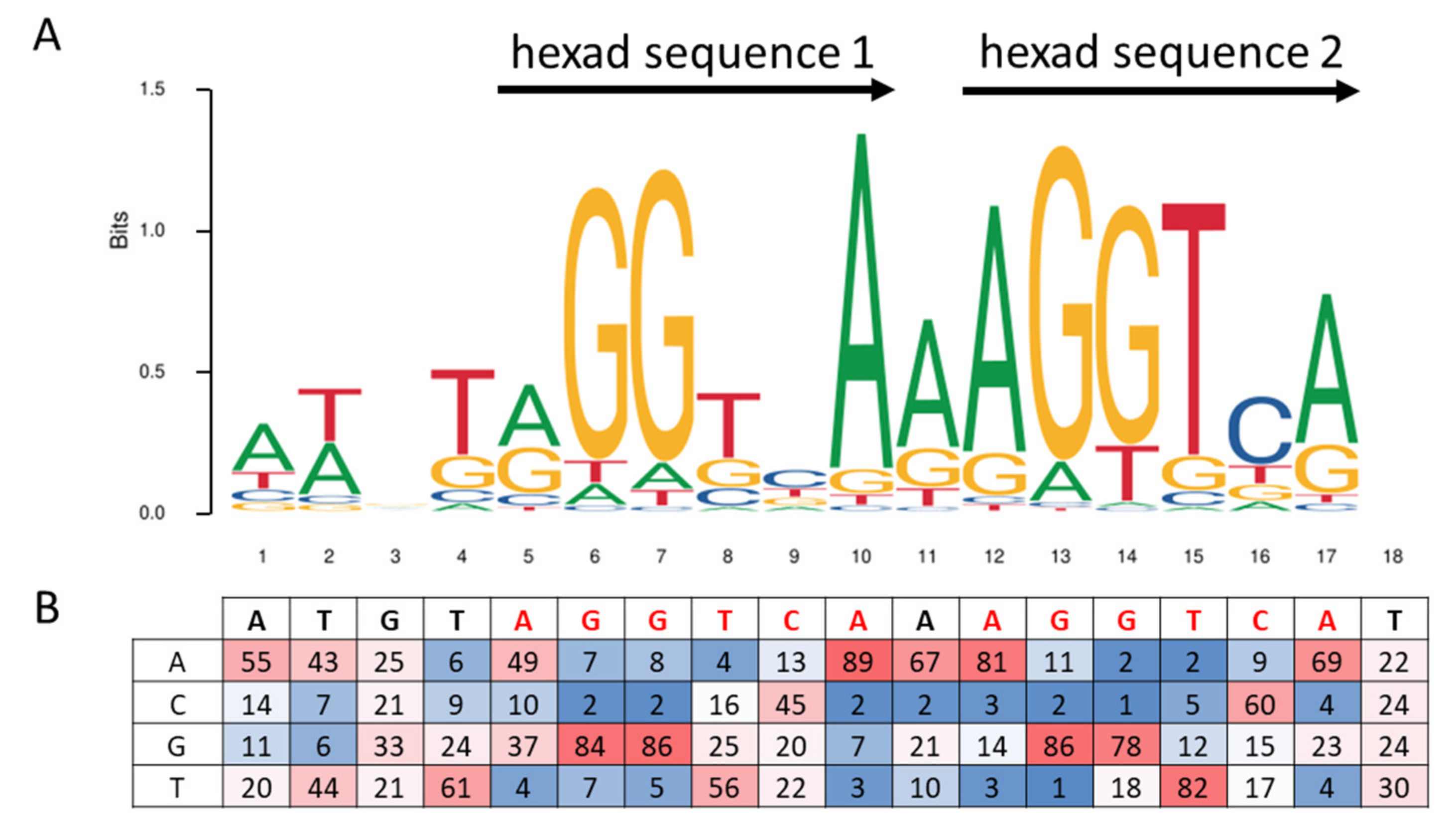
IJMS | Free Full-Text | Mechanisms Mediating the Regulation of Peroxisomal Fatty Acid Beta-Oxidation by PPARα | HTML
![1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems - ScienceDirect 1,5-Hydrogen Atom Transfer/Surzur–Tanner Rearrangement: A Radical Cascade Approach for the Synthesis of 1,6-Dioxaspiro[4.5]decane and 6,8-Dioxabicyclo[3.2.1]octane Scaffolds in Carbohydrate Systems - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0022326322014803-jo1c01376_0009.jpg)




